Acids, Bases and Salts
Acids, Bases and Salts
ic current and magnetic fields are connected, the rules to determine directions of force and current, and the applications in our daily lifeSubstances around us taste sour or bitter, feel soapy, or cause a burning sensation. The science behind these properties lies in acids, bases, and salts. These are some of the most important classes of chemical compounds used in our daily life — from food and medicine to agriculture and industry. In this chapter, we’ll explore their nature, reactions, and applications.
What Are Acids and Bases?
Acids
Acids are substances that taste sour and turn blue litmus red.
They contain hydrogen and release H⁺ (hydrogen ions) when dissolved in water.
Examples:
• Hydrochloric acid (HCl)
• Sulphuric acid (H₂SO₄)
• Nitric acid (HNO₃)
• Acetic acid (CH₃COOH)
Equation:
"HCl (aq)"→"H⁺ (aq)"+"Cl⁻ (aq)"
Acids can be:
• Strong acids: completely ionize (HCl, H₂SO₄)
• Weak acids: partially ionize (CH₃COOH)
Bases
Bases are substances that taste bitter, feel soapy, and turn red litmus blue.
They release OH⁻ (hydroxide ions) when dissolved in water.
Examples:
• Sodium hydroxide (NaOH)
• Potassium hydroxide (KOH)
• Calcium hydroxide (Ca(OH)₂)
• Ammonium hydroxide (NH₄OH)
Equation:
NaOH (aq) → Na⁺ (aq) + OH⁻ (aq)
Bases that dissolve in water are called alkalis.
Indicators – Detecting Acids and Bases
Indicators are substances that change color in acids or bases. They help us identify the nature of a solution.

Natural indicators: Litmus, turmeric, red cabbage extract
Synthetic indicators: Phenolphthalein, methyl orange
Olfactory indicators: Substances whose smell changes (onion, clove, vanilla)
Example:
• Onion loses its smell in a base but not in an acid.
The Reaction of Acids and Bases with Metals

Reaction of Acids and Bases with Each Other – Neutralization
When an acid reacts with a base, they neutralize each other to form salt and water.
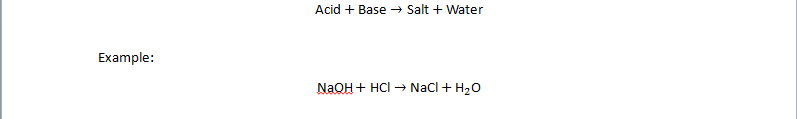
This reaction releases heat → exothermic reaction.
Neutralization plays a vital role in our body (stomach acid neutralized by antacids) and in agriculture (acidic soil neutralized by lime).
Reaction of Metal Oxides and Non-Metal Oxides
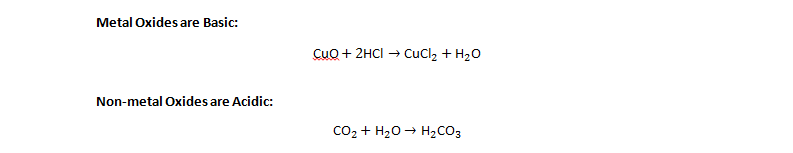
Thus, CO₂ forms carbonic acid in water → reason for acid rain.
Importance of pH in Everyday Life

Important Salts in Daily Life
(1) Common Salt – NaCl
Obtained from seawater by evaporation.
Used for cooking, preserving food, and as a raw material for other chemicals.
(2) Baking Soda – NaHCO₃ (Sodium Bicarbonate)
Prepared from brine using the Solvay process.
• In baking (releases CO₂ to make dough rise)
• As an antacid
• In fire extinguishers
Reaction while heating:
NaHCO₃ →┴( heat ) Na₂CO₃ + H₂O + CO₂
Common Acids, Bases, and Salts in Everyday Life

Crystals and Water of Crystallization
Many salts contain fixed amounts of water molecules — this is called water of crystallization.
• CuSO₄•5H₂O → Blue vitriol (5 water molecules)
• FeSO₄•7H₂O → Green vitriol
• Na₂CO₃•10H₂O → Washing soda
• CaSO₄•½H₂O → Plaster of Paris
When heated, they lose water and change color (CuSO₄•5H₂O → CuSO₄ white).
Summary of Important Concepts

Conclusion
Acids, bases, and salts form the foundation of chemistry. From the food we eat to the medicines we use, from the soil under our feet to the materials in our homes—these substances shape our world. Understanding their reactions, pH, and uses helps us apply chemistry in daily life safely and effectively.