Metals and Non-Metals
Metals and Non-Metals
Everything around us — from the iron nails in doors to the copper wires in cables — is made up of metals or non-metals. These elements are the building blocks of matter and play an important role in our daily life.
Metals are usually shiny, hard, and conduct electricity, while non-metals are dull, soft, and poor conductors.
In this chapter, we’ll explore their physical and chemical properties, how they react, and how metals are extracted and used.
Physical Properties of Metals and Non-Metals
1. Physical State
• Metals are usually solid at room temperature, except mercury (Hg) which is a liquid.
• Non-metals exist in solid, liquid, or gaseous states. Example: carbon (solid), bromine (liquid), oxygen (gas).
2. Lustre (Shininess)
• Metals have a shiny surface (metallic lustre) and can be polished.
• Non-metals are dull in appearance (except iodine which is shiny).
3. Hardness and Malleability
• Metals are generally hard and can be beaten into thin sheets (malleable).
• Gold and silver are the most malleable metals.
• Non-metals are brittle and break easily — they cannot be beaten into sheets.
4. Ductility
• Metals can be drawn into thin wires (ductile).
Example: Copper wires.
• Non-metals are not ductile.
5. Conductivity
• Metals are good conductors of heat and electricity.
(Silver and copper are best conductors.)
• Non-metals are poor conductors (except graphite, a good conductor of electricity).
6. Density and Strength
• Metals are generally dense and strong, e.g., iron, copper.
• Non-metals have low density and are soft (except diamond, the hardest natural substance).
7. Sonorous Nature
• Metals produce a ringing sound when struck (they are sonorous).
• Non-metals do not produce any sound.
8. Melting and Boiling Points
• Metals have high melting and boiling points, except mercury and gallium.
• Non-metals have low melting and boiling points.
Chemical Properties of Metals
1. Reaction with Oxygen
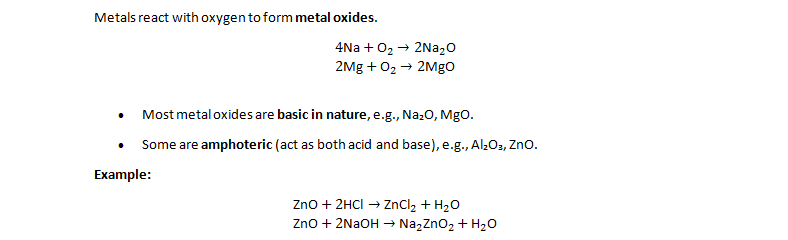
2. Reaction with Water
Different metals react differently with water.

3. Reaction with Acids
Metals react with acids to form salt and hydrogen gas.
Zn + 2HCI → ZnCi2 + H2
Metals like copper, silver, and gold do not react with dilute acids.
Hydrogen gas test: it burns with a ‘pop’ sound.
Reactivity Series
The reactivity series is a list of metals arranged in order of their reactivity from highest to lowest.

Occurrence of Metals
Occurrence of Metals:
1. Native form – uncombined state (e.g., gold, silver, platinum).
2. Combined form – as oxides, carbonates, sulfides, halides, etc. (e.g., iron oxide, zinc sulfide).
Minerals: Natural substances containing metals.
Ores: Minerals from which metals can be extracted profitably.
Purification of Metals
The extracted metal often contains impurities and is purified by electrolytic refining.

Prevention of Corrosion:
• Painting / Oiling / Greasing – prevents contact with air and water.
• Galvanization – coating iron with zinc.
• Electroplating – coating with another metal (e.g., silver or chromium).
• Alloying – mixing metals for better properties.
Uses of Metals and Non-Metals
Metals:
• Iron – construction, machinery
• Aluminium – utensils, airplanes
• Copper – electrical wires
• Silver and gold – jewellery, coins
• Zinc – galvanization
Non-Metals:
• Oxygen – respiration
• Nitrogen – fertilizers
• Sulphur – gunpowder, medicines
• Carbon – fuels, steel industry
• Iodine – antiseptics
Summary of Key Concepts

Conclusion
Metals and non-metals are fundamental to our modern world — from electrical circuits to fertilizers, they form the backbone of industry and life. Understanding their physical and chemical behaviors helps in material selection, corrosion prevention, and developing stronger, more efficient alloys for human progress.